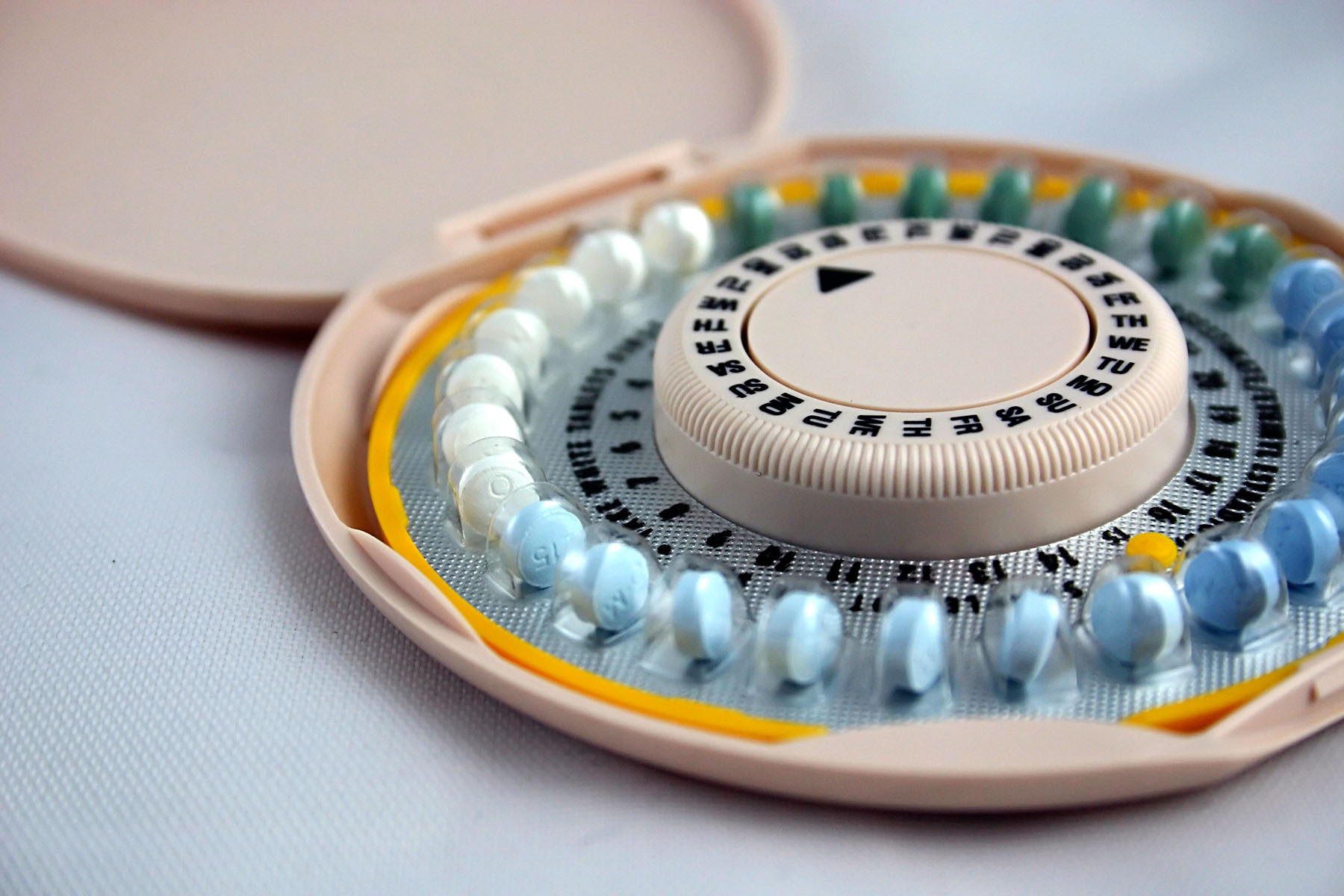
First OTC Contraceptive Pill Approved
I remember working part-time as a pharmacist when I was at FDA and there were only a few prescription oral contraceptives that were available for dispensing at that time. If you had asked then (1984) if I thought that there would ever be an OTC version of an oral contraceptive available, I would have bet […]












